On July 9st 2024, Gut (IF=23), a leading international journal in gastroenterology and hepatology, published online a research paper titled " Glutamine metabolic competition drives immunosuppressive reprogramming of intratumour GPR109A+ myeloid cells to promote liver cancer progression ", by the joint team of Prof. Hui Wang and Dr. Xiaoguang Li from School of Public Health of Shanghai Jiao Tong University, which identified a nutrient competition between liver cancer cells and myeloid cells within the tumor microenvironment. This research holds significant implications for public health, early screening of liver cancer, and precision cancer therapy. The discovery offers a novel immunometabolic checkpoint, GPR109A, for early liver cancer screening. Targeting of GPR109A+ myeloid cells at the cellular level can enhance immune cytotoxicity, thereby inhibiting the progression of liver cancer. Our findings will advance the development of individualized immunotherapeutic strategies for liver cancer based on targeting tumor-associated myeloid cells, filling the research gap on how nutritional metabolic reprogramming regulates the molecular mechanisms of liver cancer progression.

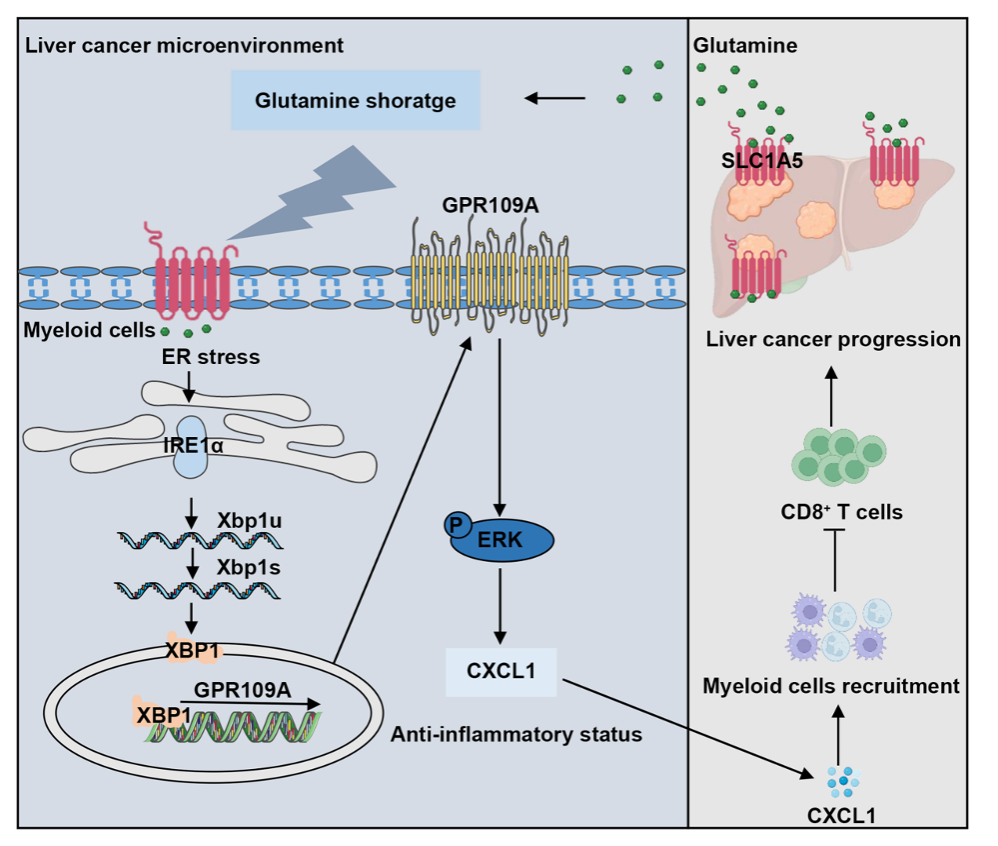
The metabolic characteristics of liver cancer drive considerable hurdles to immune cell function and cancer immunotherapy. However, how metabolic reprograming in the tumor microenvironment impairs the anti-tumor immune response is unclear. Here, we demonstrate that glutamine shortage in liver cancer tumor microenvironment drives an immunosuppressive GPR109A+ myeloid cells infiltration in both murine models and humans, leading to the evasion of immune surveillance. Genetic knockout or pharmacological blockade of GPR109A decreases G-MDSCs and M2-like TAMs abundance to trigger the anti-tumor responses of CD8+ T cells and further improves the immunotherapy efficacy against liver cancer. Mechanistically, tumor cells and tumor-infiltrated myeloid cells compete for glutamine uptake via the transporter SLC1A5 to control anti-tumor immunity, which disrupts the endoplasmic reticulum (ER) homeostasis and induces unfolded protein response of myeloid cells to promote GPR109A expression through IRE1α/XBP1 pathway. In both in vivo and in vitro studies, the restricting of glutamine uptake in liver cancer cells, as well as the blockade of IRE1α/XBP1 signaling or glutamine supplementation can eliminate the immunosuppressive effects by GPR109A+ myeloid cells and slow down tumor progression. Our findings identify the immunometabolic crosstalk between liver cancer cell and myeloid cell facilitates tumor progression via a glutamine metabolism/ER stress/GPR109A axis, suggesting that GPR109A can be exploited as an immunometabolic checkpoint and putative target for cancer treatment.
This research was highlighted and received a specialized commentary entitled “The Q for immune evasion in HCC: ER stress in myeloid cells” by Gut. Dr. Yang Yang and MS. Tianduo Pei are the first authors. Prof. Hui wang and Xiaoguang Li are the co-corresponding author. The research was supported by the National Natural Science Foundation of China, the National Key R&D Program of China, the Science and Technology Commission of Shanghai Municipality, Natural Science Foundation of Shanghai and the Innovative research team of high-level local universities.

Original link:
https://gut.bmj.com/content/early/2024/07/09/gutjnl-2024-332429
https://gut.bmj.com/content/early/2024/08/29/gutjnl-2024-333249




