On January 8th, 2025, Dr. Ningning Liu, was invited to publish a preview article titled “Fungal Influence: The Role of the Gut Mycobiome in Women’s Health” in the prestigious journal Cell Host & Microbe.The human gut microbiome is closely related to human health and diseases. In recent years, there has been unprecedented attention given to the role of the fungal microbiome in human disease development and progression, compared to bacteria. However, the causal relationship remains unclear. In addition to being harmless symbionts, fungi can also transform into pathogens in individuals with endocrine and metabolic disorders. Therefore, in-depth investigation on disease-specific fungal microbiome holds promise for developing new therapeutic strategies.

Polycystic Ovary Syndrome (PCOS) is a common condition among women worldwide, affecting approximately 11-13% of the population. It is characterized by elevated androgen levels, menstrual irregularities, and polycystic ovaries. Although existing studies have linked gut bacterial dysbiosis with the onset and progression of PCOS, the underlying mechanisms remain insufficiently understood. Particularly, the research on the role of fungal-host interactions in PCOS pathogenesis remains limited. This article explores the new study on the role of Aspergillus tubingensis (A. tubingensis) in promoting a PCOS-like phenotype. Through extensive experiments, the research team led by Dr. Yanli Pang and Professor Jie Qiao at Peking University Third Hospital found that A. tubingensis inhibited the AhR signaling pathway in ILC3 cells, reducing IL-22 secretion, and identified a fungal metabolite called AT-C1, which acts as an endogenous AhR antagonist. This metabolite induces the PCOS-like phenotype via the ILC3-AhR pathway. A. tubingensis was detected in three independent cohorts of PCOS patients in China, suggesting its potential role in PCOS development.
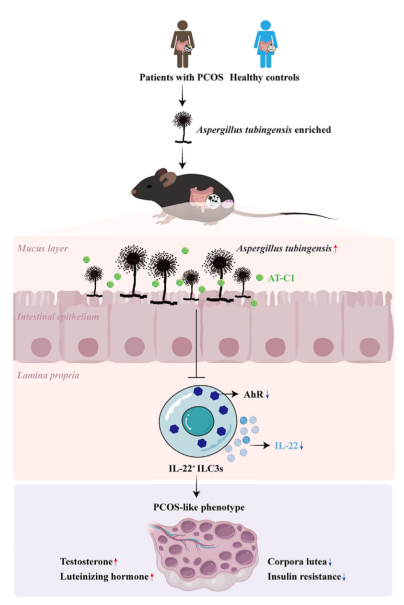
Figure 1: A. tubingensis induces a PCOS-like phenotype by inhibiting IL-22 expression through its secondary metabolites.
Future research is required to further understand the secretion mechanism of AT-C1 and its role in inducing the PCOS phenotype. Given the importance of A. tubingensis in patients with lung diseases, investigating its colonization mechanisms in the human respiratory and intestinal tracts is critical. The potential impact of A. tubingensis on the gut microbiome, as well as its regulation on AT-C1 secretion, remains an open question. Exploring A. tubingensis and its metabolite AT-C1 may help improve clinical diagnostic methods and treatment strategies for PCOS.
The first author was the graduate student Tianyi Zhang from the Center for Single-Cell Omics, School of Public Health, Shanghai Jiao Tong University School of Medicine, with Dr. Ningning Liu as the corresponding author. This preview was supported by the MOST Key R&D Program of China and the National Natural Science Foundation of China.
The article is available at: https://doi.org/10.1016/j.chom.2024.12.005

Dr. Ningning Liu is Deputy Director of Center for Single-Cell Omics in Shanghai Jiao Tong University School of Medicine. His research focuses on fungal infections and cancer progression, by integrating microbiome and single-cell omics to explore the fungi-host interactions during cancer development.

Tianyi Zhang is a master student at the State Key Laboratory of Systems Medicine for Cancer, Center for Single-Cell Omics, School of Public Health, Shanghai Jiao Tong University School of Medicine. Since joining the lab in September 2023, she has published a first-authored paper in Nature Communications and was awarded the National Scholarship for Master Student in 2024.




